Application of Thermodynamics: Heat Engine, Heat pump and Refrigeration | Working |
Thermodynamics has a wide range of applications in various fields, including physics, chemistry, engineering, biology, and material science. Here are some of the key applications of thermodynamics- heat engine, heat pump and refrigeration.
Heat engine:
In thermodynamics we use heat energy and convert it into mechanical energy. Heat is absorbed from a high-temperature reservoir and some of it is turned into work and the rest of the heat is discarded into a heat sink.
This means W =Q2-Q1, where Q2=input heat.
Q1=rejected heat after work.


Heat pump:
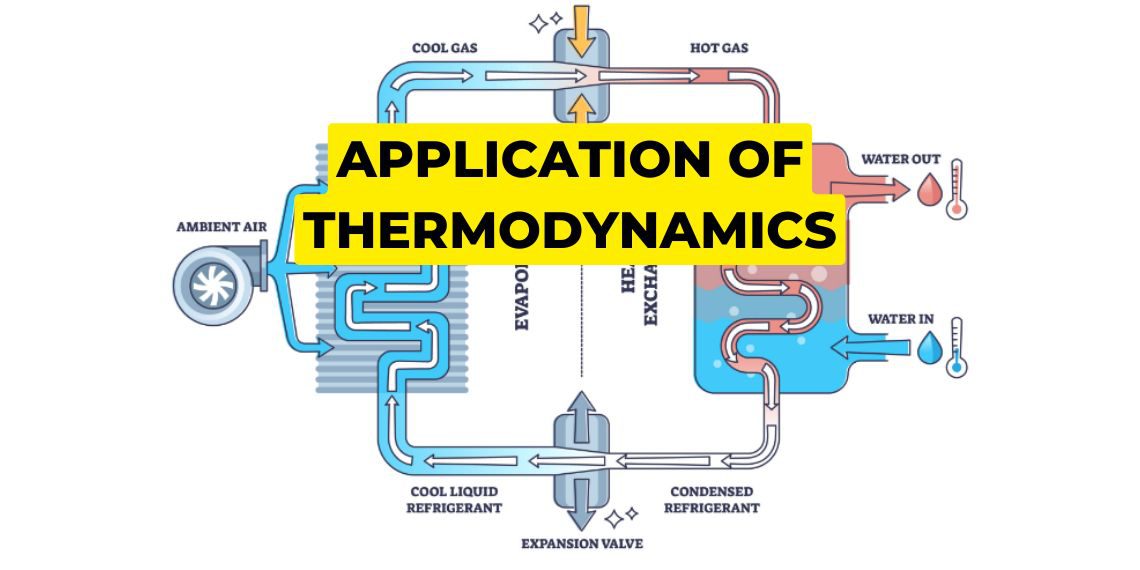
Heat pump is a mechanical device that is used to remove heat like an air conditioning system. Here our electric power is converted into mechanical energy and heat is taken from a high-temperature region and is released in the surroundings.
C.O.P =Q2/W =Q2/ Q2-Q1.
C.O.P IS ALWAYS >1.

Refrigeration:
Refrigerator system is similar to a heat pump but it works over a closed area or system. Here refrigerant is used and it will run in a cycle and this refrigerant will take the heat from inside and it goes through some process and in this process heat is rejected into the surroundings. The cycle runs continuously and decreases the temperature inside.
C.O.P =Q1/W
=Q1/Q2-Q1
C.O.P CAN BE >1, <1, OR =1.

Note:
If you want to learn more about this topic, we suggest checking out our Combo package with the given link https://merchantnavydecoded.com/courses/c/ . It’s a great way to dive deeper into the subject through video explanations. This package covers all the important details and presents them in an easy-to-understand format. Watching the videos will help you grasp the topic better and make learning more enjoyable. So, we highly recommend giving our Combo package a try to enhance your knowledge on the subject.
Disclaimer :- The opinions expressed in this article belong solely to the author and may not necessarily reflect those of Merchant Navy Decoded. We cannot guarantee the accuracy of the information provided and disclaim any responsibility for it. Data and visuals used are sourced from publicly available information and may not be authenticated by any regulatory body. Reviews and comments appearing on our blogs represent the opinions of individuals and do not necessarily reflect the views of Merchant Navy Decoded. We are not responsible for any loss or damage resulting from reliance on these reviews or comments.
Reproduction, copying, sharing, or use of the article or images in any form is strictly prohibited without prior permission from both the author and Merchant Navy Decoded.